<< Industry news

Lumos Diagnostics has today announced that U.S. FDA has granted 510(k) clearance with CLIA waiver for FebriDx®, enabling broader use across frontline healthcare settings and significantly expanding its commercial opportunity.

Roche announced today the U.S. Food and Drug Administration (FDA) 510(k) clearance of its newest analytical units – the cobas® c 703 and cobas® ISE neo. As part of the scalable and modular cobas® pro integrated solutions, these additions deliver advanced lab-automation capabilities designed to help address some of today's most pressing laboratory challenges, including staff shortages, limited space and growing test volumes.

Amoy Diagnostics Co., Ltd., Medical & Biological Laboratories Co., Ltd., a group company of Tokuyama Corporation, and Precision Medicine Asia Co., Ltd. today announced that MBL received approval from the Japanese Ministry of Health, Labour and Welfare (MHLW) on March 5, 2026 for the AmoyDx® PIK3CA Mutation Detection Kit in Japan. The approval in Japan marks an important milestone achieved through the close collaboration of MBL, AmoyDx and PREMIA.

Roche (SIX: RO, ROP; OTCQX: RHHBY) announced today the launch of the cobas® eplex respiratory pathogen panel 3 (RP3), a new diagnostic test designed to detect a broad spectrum of viruses and bacteria that cause respiratory illness. The test is now available in countries accepting the CE mark.

Themed "Global IVD Innovation & Cross-Regional Integration," this groundbreaking event brings together distinguished experts from around the world to explore the future of diagnostics.
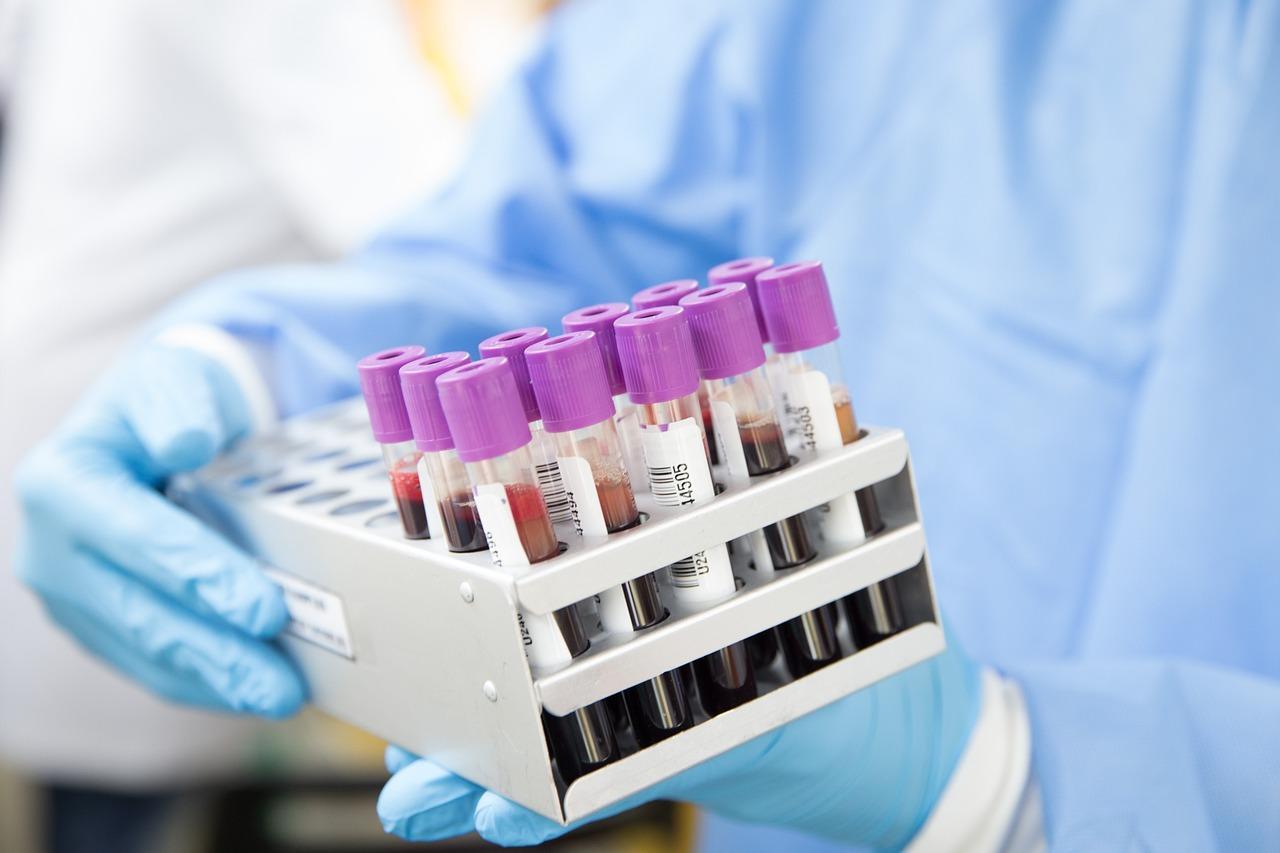
Blood typing reagents mainly refer to reagents and kits used separately or in combination with blood grouping analyzer, which belong to a segment of in vitro diagnostic (IVD) reagent industry.

PathAI, Inc., a global leader in artificial intelligence (AI)-powered pathology solutions, today announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation to PathAssist Derm1, designed to analyze digital pathology whole slide images (WSIs) of skin lesions and aid pathologists in their review.

Quanterix Corporation (NASDAQ: QTRX), a company fueling scientific discovery through ultra-sensitive biomarker detection, today announced financial results for the fourth quarter and full year ended December 31, 2025.

SOPHiA GENETICS (Nasdaq: SOPH), a global leader in AI-driven precision medicine, today provided preliminary unaudited financial results for the fourth quarter and full year 2025, initiated its financial outlook for 2026, and announced an executive transition plan, including the promotion of Ross Muken to Chief Executive Officer (CEO), effective July 1, 2026, and the transition of co-Founder Jurgi Camblong to Executive Chairman.

Natera, Inc. (NASDAQ: NTRA), a global leader in cell-free DNA and genetic testing, today reported its financial results for the fourth quarter and full year ended December 31, 2025.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
