


Moderator: Irene Aihie April 22, 2020 12:15 pm ET Coordinator: Welcome and thank you for standing by. At this time all parties are in a listen-only mode until the question and answer segment of todays conference at which time you may press
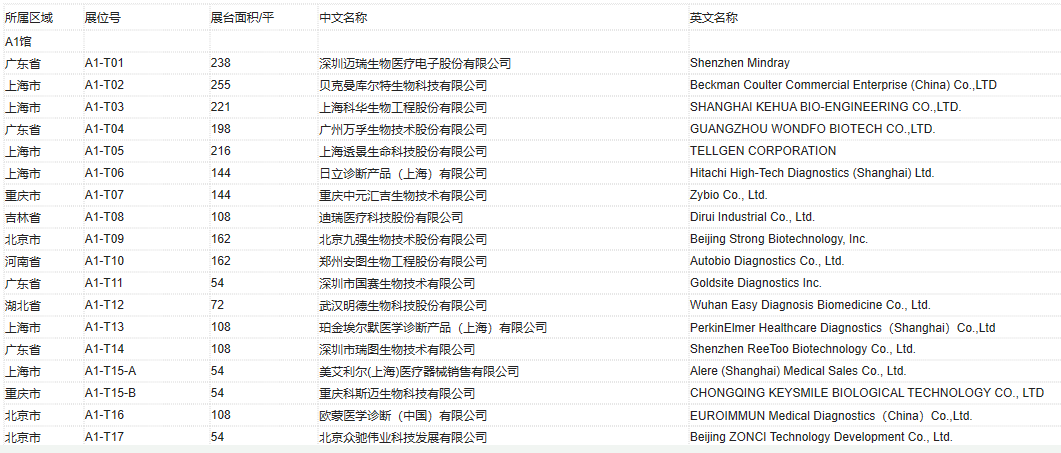
2020 CACLP Expo Exhibitor List
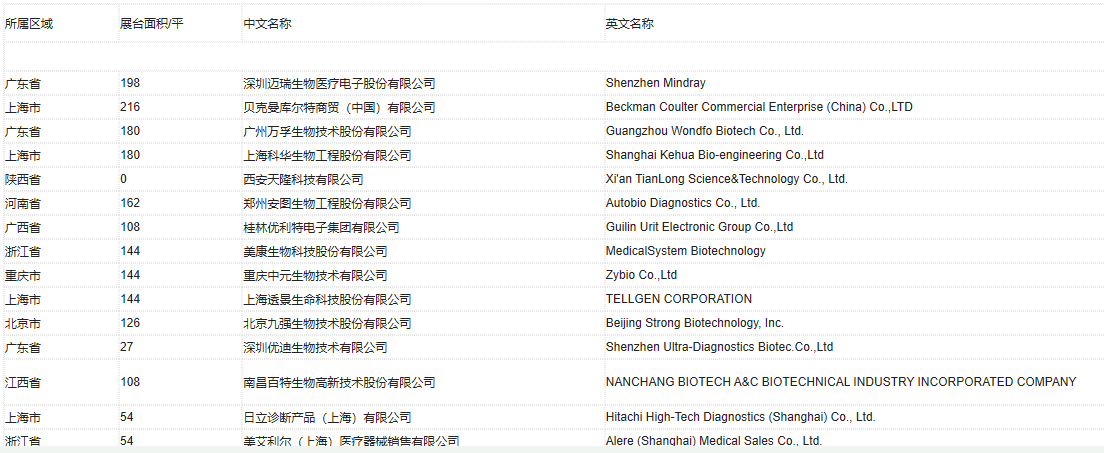
2019 CACLP Expo Exhibitor List
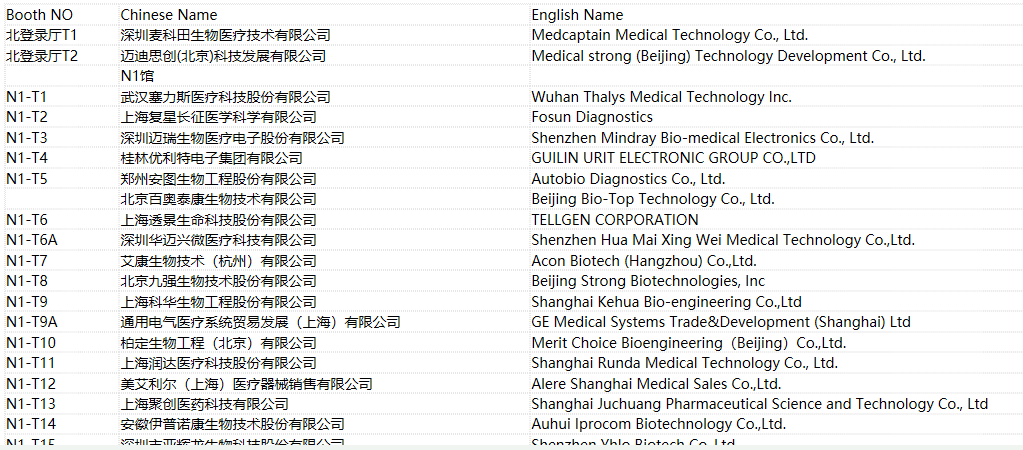
2018 CACLP Expo Exhibitor List

2017 CACLP Expo Exhibitors List

2020 CACLP Expo Exhibitor List

2019 CACLP Expo Exhibitor List

2018 CACLP Expo Exhibitor List

2017 CACLP Expo Exhibitors List

16TH CACLP
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
