


To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

ARUP Laboratories today announced that the U.S. Food and Drug Administration (FDA) has approved AAV5 DetectCDx™ as a companion diagnostic to aid in the selection of adult patients eligible for treatment with ROCTAVIAN™ (valoctocogene roxaparvovec-rvox).

Based on the advantages of their respective fields, the two companies will carry out in-depth cooperation in life information and support, in vitro diagnosis, medical imaging, overall solutions for two cancer screening, hospital department equipment, information construction, etc., and jointly establish the "Hybribio-Mindray Standardized Laboratory" to achieve complementary advantages and resource sharing.

Bio-Techne announced on Thursday that it is acquiring Swiss spatial biology firm Lunaphore for an undisclosed amount.

PamGene International said Tuesday that the European Innovation Council's Accelerator program will provide €7.5 million ($8.2 million) in funding to develop and market the firm's prognostic test for immunotherapy response in metastatic cancer treatment.
In some areas with weak medical resources in China, it is difficult for the public to obtain quality medical services and have to seek treatment across provinces and regions, which is a heavy economic and time burden.

MOROCCO – Techniques Science Santé (T2S), a distributor of high-tech medical equipment based in Casablanca, has expanded a multi-year agreement with American company GE Healthcare, with a focus on ultra-modern modern technology.

Thermo Fisher Scientific Inc., the world leader in serving science, has completed its acquisition of MarqMetrix, a privately held developer of Raman-based spectroscopy solutions for in-line measurement. The terms of the deal were not disclosed.

Roche said Tuesday that it secured US Food and Drug Administration 510(k) clearances for a pair of assays to aid diagnosis of Alzheimer's disease measuring levels of beta-amyloid and tau proteins.
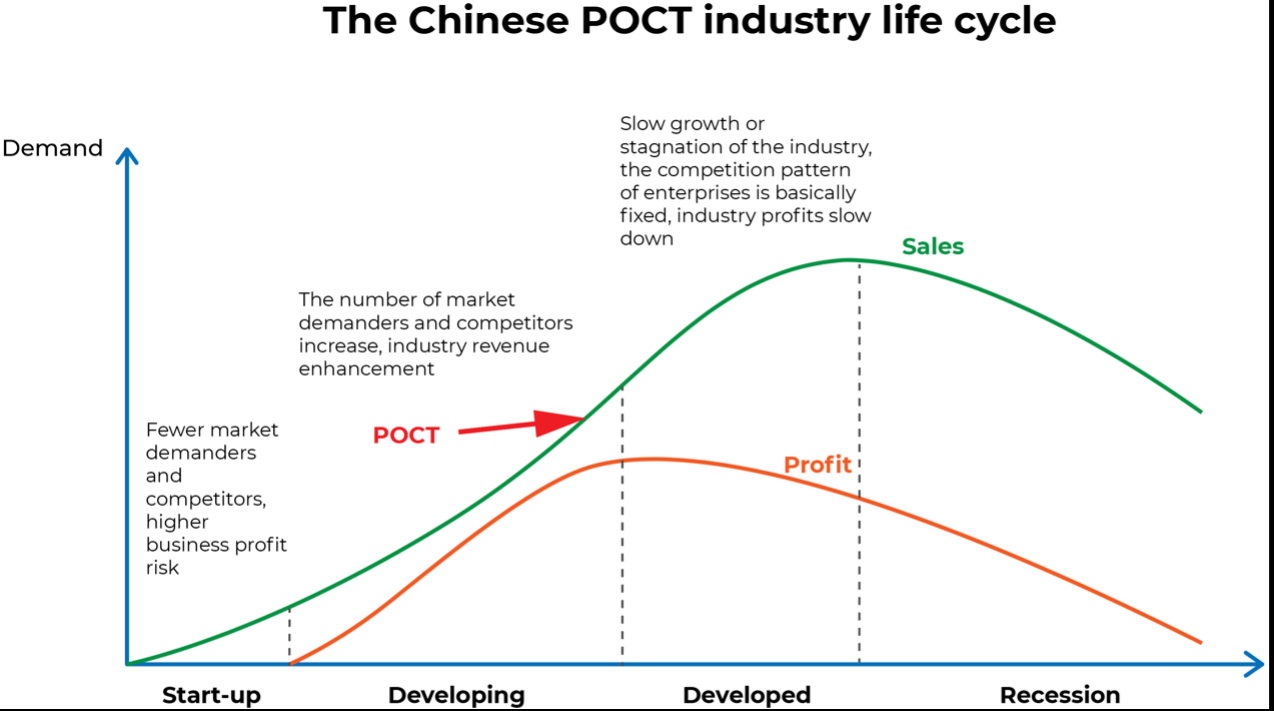
POCT industry started late in China, the overall market size is still small currently, and the penetration rate in hospitals and other terminal sites is low, so there is a huge potential development space. Qianzhan Institute predicts that from 2022-2027, the Chinese POCT market will rise by about 9% CAGR to about 18 billion yuan in 2027.

Prenetics Global Limited, a leading genomics-driven health sciences company, and Prof. Dennis Lo today announce they have entered into an agreement to establish a joint venture named Insighta. The board of directors of Prenetics Global Limited has unanimously approved the transaction.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
