


To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.
The US Food and Drug Administration (FDA) has granted approval for Lumos Diagnostics’ FebriDx rapid point-of-care (POC) test.

Today, in collaboration with single cell genomics company, Honeycomb Biotechnologies (“Honeycomb”), Revvity (NYSE: RVTY) announced the launch of a suite of new solutions and services that expand the frontiers of single cell biology: the HIVE™ CLX Single-Cell RNAseq Solution, BeeNetPLUS analysis workflow, and for single cell researchers based in the United States, a new HIVE CLX Service offering.
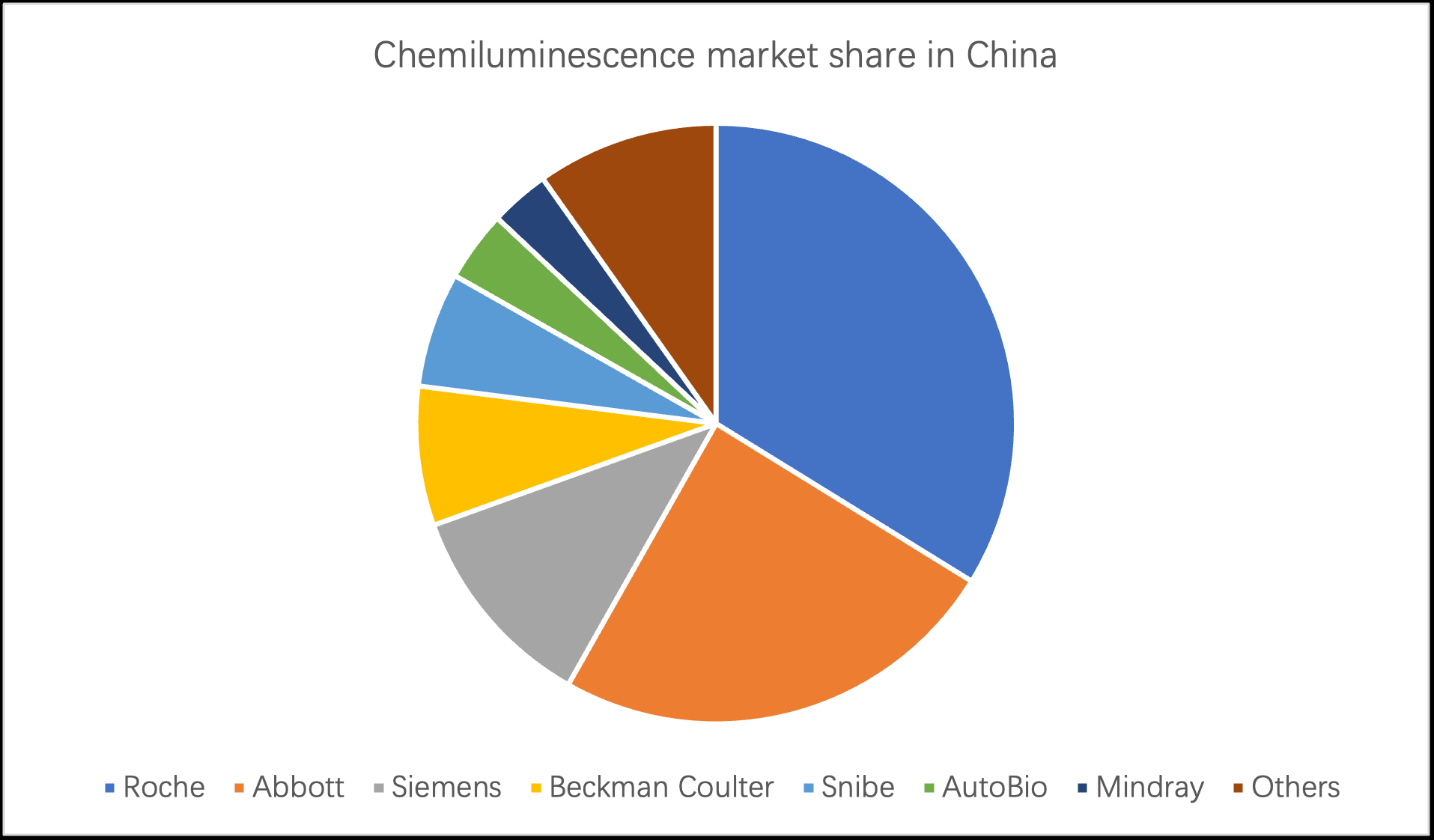
In recent years, the market scale of immunodiagnostic reagents in China has been increasing year by year. In 2020, the market size of China's immunodiagnostic reagent industry reached 32.1 billion yuan, an increase of 24.42% year-on-year.

Renalytix has received De Novo marketing authorisation from the US Food and Drug Administration (FDA) for its KidneyIntelX.dkd prognostic test.

Houston-based single-cell analysis company CellChorus announced on Wednesday that it has been awarded a $2.2 million Small Business Innovation Research (SBIR) Fast-Track grant from the National Institute of General Medical Sciences (NIGMS).

On June 29, Merck announced an investment of approximately 70 million euros to expand its Nantong Life Science Center in the heart of the Yangtze River Delta region.

LumiraDx Limited (Nasdaq: LMDX), a leading innovator in diagnostic technologies, today announced the submission of its first 510(k) application to the U.S. Food and Drug Administration (FDA) for the clearance of its ground-breaking 5-minute COVID Ultra Test. LumiraDx submitted a traditional 510(k) Dual Submission for LumiraDx SARS-CoV-2 Ag Ultra with CLIA Waiver for the commercial distribution at point of care of the LumiraDx Platform, including the LumiraDx Instrument, with the LumiraDx SARS-CoV-2 Antigen (Ag) Ultra test, and the LumiraDx SARS-CoV-2 Ag Quality Control Swab Kit.

Helio Genomics said Thursday that its sister company, Laboratory for Advanced Medicine & Health Group (LAMH), has received approval from the National Medical Products Administration (NMPA) in China for its cell-free DNA-based liver cancer detection test. The assay is the first of its kind to receive this approval.

At 9:30 a.m. on June 30, Adicon was officially listed on the Hong Kong Stock Exchange. Adicon Holdings (09860.HK) is offering 33,192,500 shares globally, of which 3,320,000 shares are offered in Hong Kong, China and 29,872,500 shares are offered internationally, with a 15% over-allotment option.

Eli Lilly and Company (NYSE: LLY) and Sigilon Therapeutics, Inc. (Nasdaq: SGTX) today announced a definitive agreement for Lilly to acquire Sigilon, a biopharmaceutical company that seeks to develop functional cures for patients with a broad range of acute and chronic diseases.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
