Original from: bioMérieux
- First quarter was impacted by mild respiratory epidemiology, and by the uncertain geopolitical and macroeconomic environment which drives softness in customers’investment decisions.
- -3.9% organic sales decline in Q1 2026, reflecting a weaker respiratory season compared with the exceptionally strong epidemiology in Q1 2025 (resulting in BIOFIRE®1 respiratory panels sales down 23%) and a slow start of the year in instruments sales (-18%) in a soft market for new installations.
- GO•28 growth drivers’ reagents sales up 6.5% organically, notably +31% in SPOTFIRE®2 reagents sales and +8% in industrial applications reagents sales.
- Considering the first-quarter performance and the current market dynamics, bioMérieux revises its 2026 full-year guidance:
o Annual sales are now expected to grow organically between +3% to +5% (versus between +5% and +7% previously);
o Annual CEBIT3 is expected to grow organically between 0% and 10% (versus at least +10% previously) excluding a prolonged impact of Middle East events.
- Confirmation of a full-year CEBIT headwind from currency effect3 of -€50m to €60m.
Pierre Boulud, Chief Executive Officer, said: “In Q1 26, we continued to deliver on innovation, with the launch of SPOTFIRE® mini assays in Europe, the regulatory filing of SpinChip Hs-troponin assay, the launch of Mycoplasma assay on SPOTFIRE®, and the acquisition of Accellix for Pharma Quality Control. For 2026, acknowledging the particularly soft market environment that weighed on first-quarter performance, bioMérieux has revised its full-year guidance downward. Leveraging our GO•28 initiatives, we will maintain strict and disciplined control of our cost base, to ensure a positive organic evolution of CEBIT in 2026."
bioMérieux, a world leader in the field of in vitro diagnostics, today releases its business review for the three months ended March 31, 2026.
SALES
Note: Unless otherwise stated, growth is expressed year-on-year at constant exchange rates and scope of consolidation (like-for-like).
Consolidated sales totaled €984 million in the first quarter of 2026, down 10.4% as reported from €1,098 million in first quarter of 2025. Organic growth (at constant exchange rates and scope of consolidation) stood at -3.9%. Currency effect had a negative impact of €73 million over the first quarter sales, mainly due to the depreciation of the US dollar against the euro.

ANALYSIS OF SALES BY APPLICATION
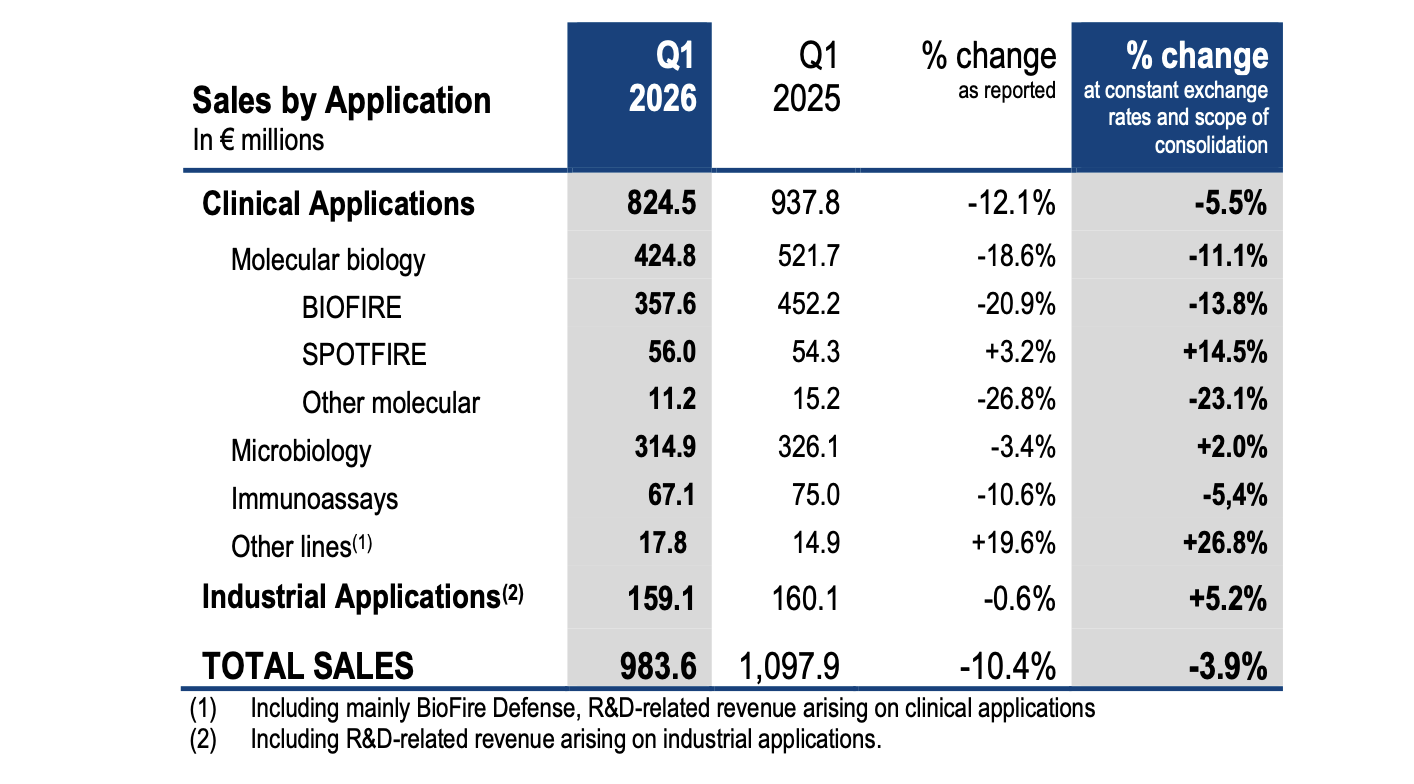
Q1 2026 vs Q1 2025 like-for-like trends per application:
- Clinical applications sales (84% of total sales), decreased by 5% to €825 million in the first quarter of 2026.
▪ In molecular biology:
o BIOFIRE® respiratory panels sales declined by 23%, mainly reflecting lower testing volumes due to a low respiratory epidemiology, compared with very strong respiratory activity in Q1 2025.
o BIOFIRE® non-respiratory panels delivered 4% sales growth, driven by double-digit growth in Blood Culture Identification (BCID) and Joint Infection (JI) panels sales. This was partly offset by a softer-than-expected performance of Pneumonia (-2%, reflecting the very mild respiratory season) and Meningitis panels, while the installed base continued to expand during the quarter.
o SPOTFIRE® sales reached €56m, up 15%, despite the very low respiratory season compared with the 1st quarter of 2025. Reagents sales increased by 31% while instrument sales were negatively impacted by a high comparison base in Japan where Q1 2025 benefited from a one-time public funding scheme. The installed base grew by 450 instruments, exceeding 6,800 units at the end of March 2026.
▪ In microbiology, sales increased by 2% in line with expectations. Quarterly performance was still impacted by activity in China (-7%) and a soft performance in equipment sales in a challenging environment that tends to delay customers’ investment decisions. Excluding China, combined BACT/ALERT® and VITEK® reagent sales grew mid-single digit.
▪ In immunoassays, sales declined by 5%, continuing to be impacted by the decrease in global VIDAS® PCT (Procalcitonin) sales and a challenging market environment in China.
▪ Industrial applications sales (16% of total sales) grew by 5%, supported by high single-digit growth in reagents sales, notably in blood culture, cytometry and molecular ranges. This growth was partly offset by a decline in instruments sales which reflects some customers’ prudence.
ANALYSIS OF SALES BY REGION
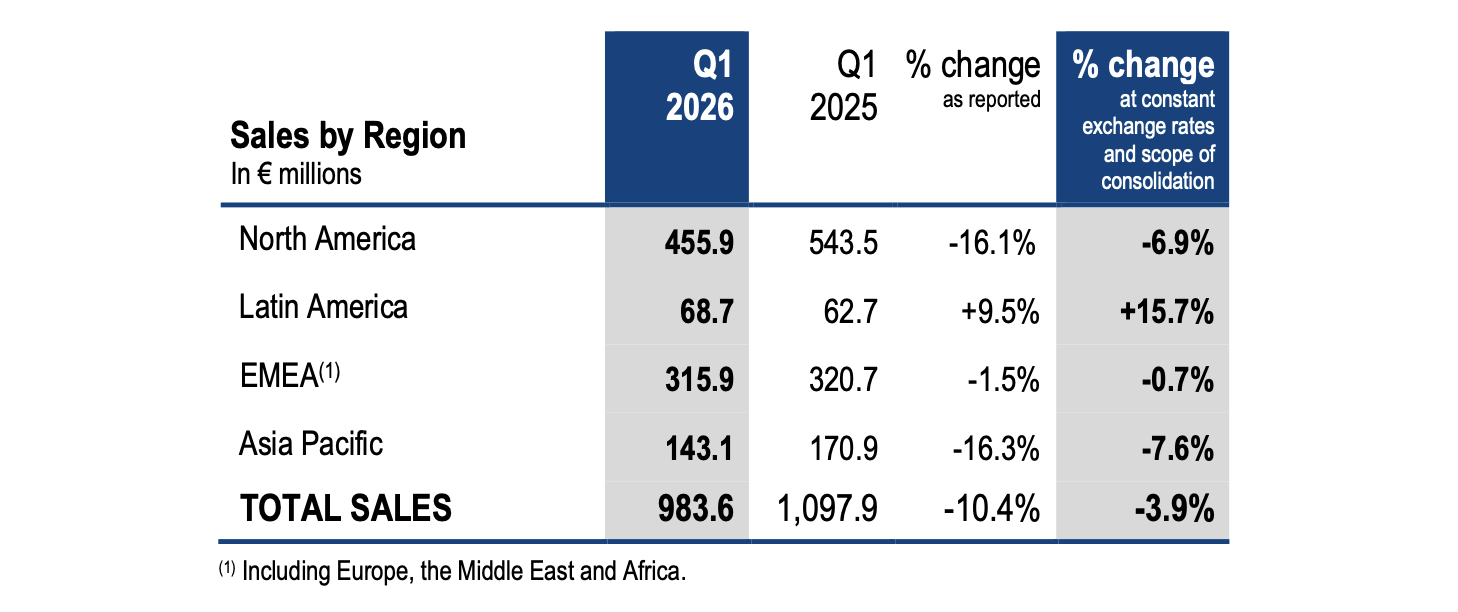
Q1 2026 vs Q1 2025 like-for-like trends per region:
- In North America (46% of total sales), revenue decreased by 7% impacted by the setback of BIOFIRE® respiratory panels sales that more than offset the double-digit growth in Industrial Applications sales and the close to 30% increase in SPOTFIRE® sales.
- In Latin America (7% of total sales), sales grew +16%, driven by strong molecular sales (+22%) and a double-digit growth in microbiology sales.
- Sales in the Europe – Middle East – Africa region (32% of total sales) totaled €316 million, a slight 1% decline. The high single digit sales growth in blood culture and in molecular biology (excluding BIOFIRE® respiratory panels) were almost entirely offset by the decline in BIOFIRE® respiratory sales.
- Sales in the Asia Pacific region (15% of total sales) decreased by 8%. The double-digit sales growth in India and the close to 50% increase in SPOTFIRE® reagents sales in Japan were compensated by the expected strong decline in SPOTFIRE® instrument sales in Japan (boosted in Q1 2025 by a special government funding scheme), as well as the decrease in BIOFIRE® respiratory panels sales across the region and a mid-single digit decline in sales in microbiology in China, still impacted by downward pressure on local healthcare spend in hospitals.
EVENTS OF FIRST QUARTER 2026
- bioMérieux receives IVDR CE Marking for two SPOTFIRE® panels to strengthen near-patient
respiratory and sore throat diagnostics across Europe
In March 2026, bioMérieux announced that its BIOFIRE® SPOTFIRE® Respiratory/Sore Throat plus (R/STplus) Panel and BIOFIRE® SPOTFIRE® Respiratory/Sore Throat plus (R/STplus) Panel Mini have been CE-marked under the IVDR. The certification includes conformity assessment for near-patient testing, enabling its use outside traditional laboratories, directly at the point of care. These multiplex PCR tests can simultaneously detect the most common pathogens responsible for respiratory and sore throat infections, within a timeframe aligned with a typical patient visit.
- bioMérieux launches SMARTBIOME™ to help food manufacturers gain deeper insights into spoilage and protect product quality
In February 2026, bioMérieux announced the launch of SMARTBIOME™, an innovative solution designed to help food manufacturers better understand, control and prevent microbiological spoilage. By combining high‑precision DNA sequencing, advanced bioinformatics and bioMérieux’s microbiology expertise, SMARTBIOME™ provides actionable insights to protect product quality, reduce waste and better preserve consumer safety.
- bioMérieux acquires Accellix
On January 22nd, 2026 bioMérieux acquired Accellix, a US company specializing in rapid, automated flow cytometry solutions for cell and gene therapy quality control. With this strategic transaction, bioMérieux strengthens its Pharmaceutical Quality Control activity and invests in innovative solutions that will support the growing advanced therapy market and improve patient outcomes worldwide.
SUBSEQUENT EVENTS
- bioMérieux launches the BIOFIRE® SPOTFIRE® molecular testing solution for the pharmaceutical industry, redefining rapid mycoplasma testing
On April 9th, 2026, bioMérieux announced the launch of BIOFIRE® SPOTFIRE® for pharmaceutical industries. This platform introduces a simple, small and smart approach to mycoplasma detection, helping biopharmaceutical manufacturers strengthen quality control, accelerate decision-making, and protect patients.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
