

As of 30 April 2026, a total of 86 China-listed companies related to the In Vitro Diagnostics (IVD) sector had released their 2025 annual reports, including 61 companies primarily engaged in IVD businesses and 25 companies with IVD-related operations.

Stay up-to-date with the latest happenings in the rapidly evolving field of In Vitro Diagnostics (IVD) in China.
Junction, the infrastructure platform for diagnostic testing and health data workflows, today announced a collaboration with GRAIL to support scalable access to the Galleri® multi-cancer early detection test across digital health platforms and clinical organizations.

Illumina, Inc. (NASDAQ: ILMN) and Labcorp (NYSE: LH), a global leader of innovative and comprehensive laboratory services, today announced an expanded collaboration to advance precision oncology through innovative applications of next-generation sequencing (NGS) solutions across the healthcare ecosystem. Projects are expected to include promoting equitable access to cancer biomarker testing by bringing it closer to patients through new distributed test offerings; generating evidence to facilitate payer coverage; and developing new tests to address areas of unmet need.
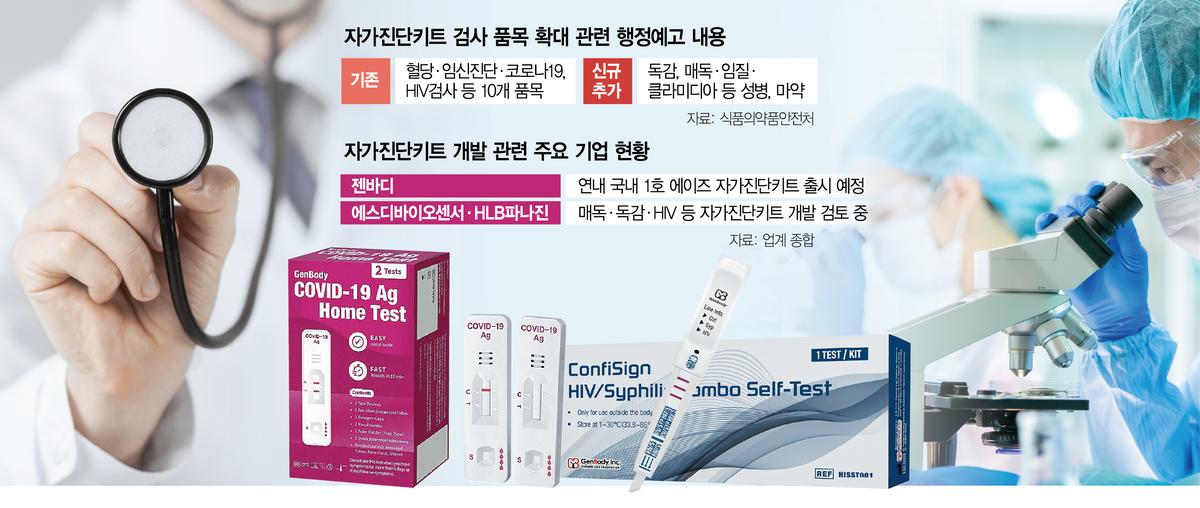
Korea's diagnostics industry is buzzing as the Ministry of Food and Drug Safety (MFDS) moves to expand the range of conditions that can be tested with home diagnostic kits to include influenza and sexually transmitted diseases. Market growth expectations are rising as diagnostic kits previously restricted to professional use become available to general consumers. Companies are accelerating development of self-testing kits in response to the policy shift.

To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

PathAI, a leading provider of AI-powered pathology solutions, and MedStar Health, a leading health system headquartered in Columbia, Maryland, today announced a multi-year strategic collaboration to deploy PathAI’s AISight® Dx1 Digital Pathology Platform and various AI algorithm products across the multi-site network.

Waters Corporation (NYSE: WAT) today announced that the U.S. Food and Drug Administration (FDA) has cleared the Onclarity HPV Self-Collection Kit and approved the BD Onclarity HPV Assay with extended genotyping for at-home use, marking a significant milestone in expanding access to cervical cancer screening, and removing barriers that currently prevent many individuals from receiving routine screening.

Hologic, Inc. (Nasdaq: HOLX), a global leader in women’s health, today announced the completion of its acquisition by funds managed by Blackstone and TPG in a transaction valued at up to $79 per share, establishing Hologic as a private company. The transaction includes significant minority investments from a wholly owned subsidiary of the Abu Dhabi Investment Authority (“ADIA”) and an affiliate of GIC. In connection with the completion of this transaction, Hologic today announced the appointment of José (Joe) E. Almeida as Chief Executive Officer, effective immediately.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Abbott (NYSE: ABT), the global healthcare company, and Flatiron Health, a leading healthtech company advancing point-of-care solutions in oncology, today announced the integration of Abbott’s comprehensive Precision Oncology portfolio into OncoEMR®, Flatiron’s cloud-based Electronic Medical Record (EMR) platform.

Co-Diagnostics, Inc. (NASDAQ: CODX) ("Co-Diagnostics," "Co-Dx," or "the Company"), a molecular diagnostics company with a unique, patented platform for the development of molecular diagnostic tests, today announced its financial results for the full year ended December 31, 2025.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
