


To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Thermo Fisher Scientific said on Wednesday that it has obtained US Food and Drug Administration clearance for its BRAHMS CgA II KRYPTOR test, a chromogranin A (CgA) immunoassay for assessing tumor progression in gastroenteropancreatic neuroendocrine tumor (GEP-NET) patients.

In 2023, NHC and 10 other departments issued the Action Plan Towards the Elimination of Cervical Cancer (2023-2030). The goal is that by 2025, HPV vaccination services for girls of school age will be promoted on a pilot basis, the cervical cancer screening rate for women of school age will reach 50%, and the treatment rate for patients with cervical cancer and precancerous lesions will reach 90%.

Cell Signaling Technology (CST), a life science discovery technology company and leading provider of antibodies, kits, and services, is partnered with Leica Biosystems, a technology leader in automated staining and brightfield and fluorescent imaging, to support the development of companion diagnostics (CDx) assays using the extensive portfolio of best-in-class antibodies from CST validated for immunohistochemistry (IHC).

Through MDUFA V, the FDA committed to several international harmonization initiatives and secured additional resources to support the work. The publication of a draft strategic plan was the first item on the MDUFA V convergence and reliance agenda.

China Association of In Vitro Diagnostics (CAIVD) and In Vitro Diagnostics Society of China Association for Medical Devices Industry jointly compiled this report.

Despite concerns from researchers and organizations, the U.S. National Institutes of Health (NIH) is adamant about implementing its new mandate requiring foreign partners to share raw data and notebooks at regular set intervals with their primary U.S. grant recipient, releasing its final guidance on September 15.

Streck announced on Monday that its MDx-Chex quality controls have received clearance from the US Food and Drug Administration.

Stay up-to-date with the latest happenings in the rapidly evolving field of In Vitro Diagnostics (IVD) in China.
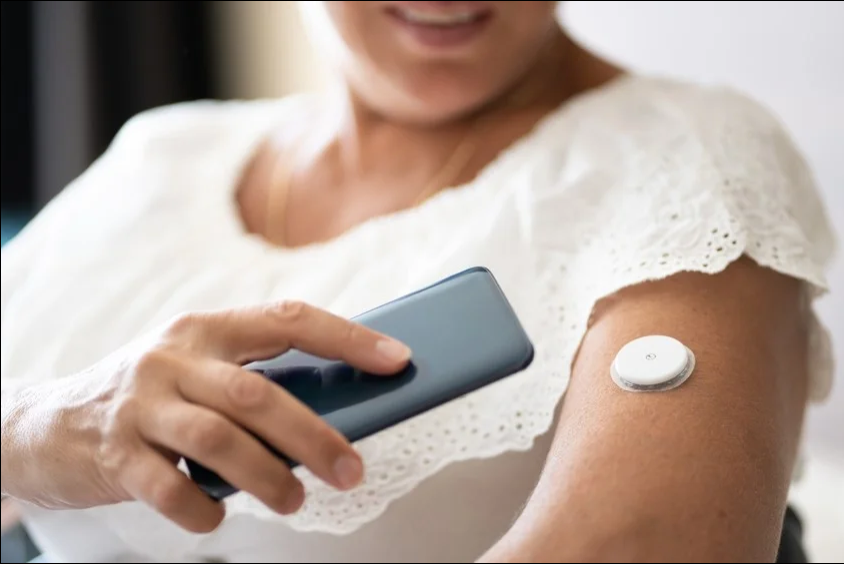
Medtronic has received a CE mark for its continuous glucose monitoring (CGM) device, Simplera, allowing the device to be marketed in the European Union.

Abbott (NYSE: ABT) today announced it has completed the acquisition of Bigfoot Biomedical, a leader in developing smart insulin management systems for people with diabetes.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
