


To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Laboratory services provider Stone Diagnostics will offer Fibronostics' artificial intelligence-based liver disease test to physicians in the US, the companies announced on Tuesday.

Fujirebio Holdings, Inc. (HQ: Minato-ku, Tokyo; President & CEO: Goki Ishikawa) and Sysmex Corporation (HQ: Kobe, Japan; President: Kaoru Asano) announce that they have entered into a Basic Agreement on Business Collaboration to deepen their multifaceted collaboration, such as on research and development, production, clinical development, and sales-marketing, in the field of immunoassay.

China Association of In Vitro Diagnostics (CAIVD) and In Vitro Diagnostics Society of China Association for Medical Devices Industry jointly compiled this report.

Invitae (NYSE: NVTA), a leading medical genetics company, today announced it gained FDA market authorization for its Common Hereditary Cancers Panel.

Foundation Medicine Inc. today announced that it has received approval from the U.S. Food and Drug Administration (FDA) for FoundationOne®CDx to be used as a companion diagnostic for Eli Lilly and Company’s Retevmo (selpercatinib), which was approved by the FDA for the treatment of adult patients with locally advanced or metastatic solid tumors with a RET gene fusion that have progressed on or following prior systemic treatment or who have no satisfactory alternative treatment options.

Stay up-to-date with the latest happenings in the rapidly evolving field of In Vitro Diagnostics (IVD) in China.

Agilent Technologies Inc. (NYSE: A) today announced the signing of a Memorandum of Understanding (MOU) with the Sarawak Infectious Disease Centre (SIDC) in East Malaysia.
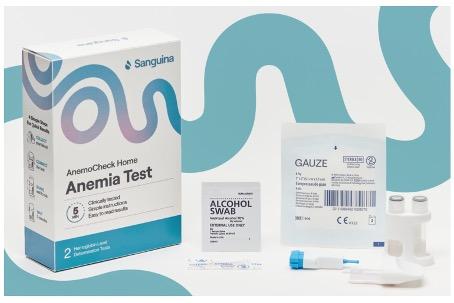
Today, Sanguina, Inc. (Sanguina), a leading biotech company, is pleased to announce the FDA clearance of AnemoCheck Home, the only FDA-cleared home hemoglobin test kit available in the United States.

China Association of In Vitro Diagnostics (CAIVD) and In Vitro Diagnostics Society of China Association for Medical Devices Industry jointly compiled this report.

Recently, Autobio's hepatitis B virus surface antigen (HBsAg) detection kit (magnetic particle chemiluminescence method) successfully passed the European clinical assessment and obtained the first EU CE IVDR Class D certification in China.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
