
Copan Diagnostics, a leader in microbiology laboratory innovations, is proud to announce that UriSponge®, a urine collection and transport device which uses a new formulation of advanced preservatives to ensure specimen stability for culture, has received clearance from the U.S. Food and Drug Administration (FDA). The solution offers clinical laboratories and healthcare providers a user-friendly, reliable, and cost-effective way to improve urine specimen collection, preservation, and transport.
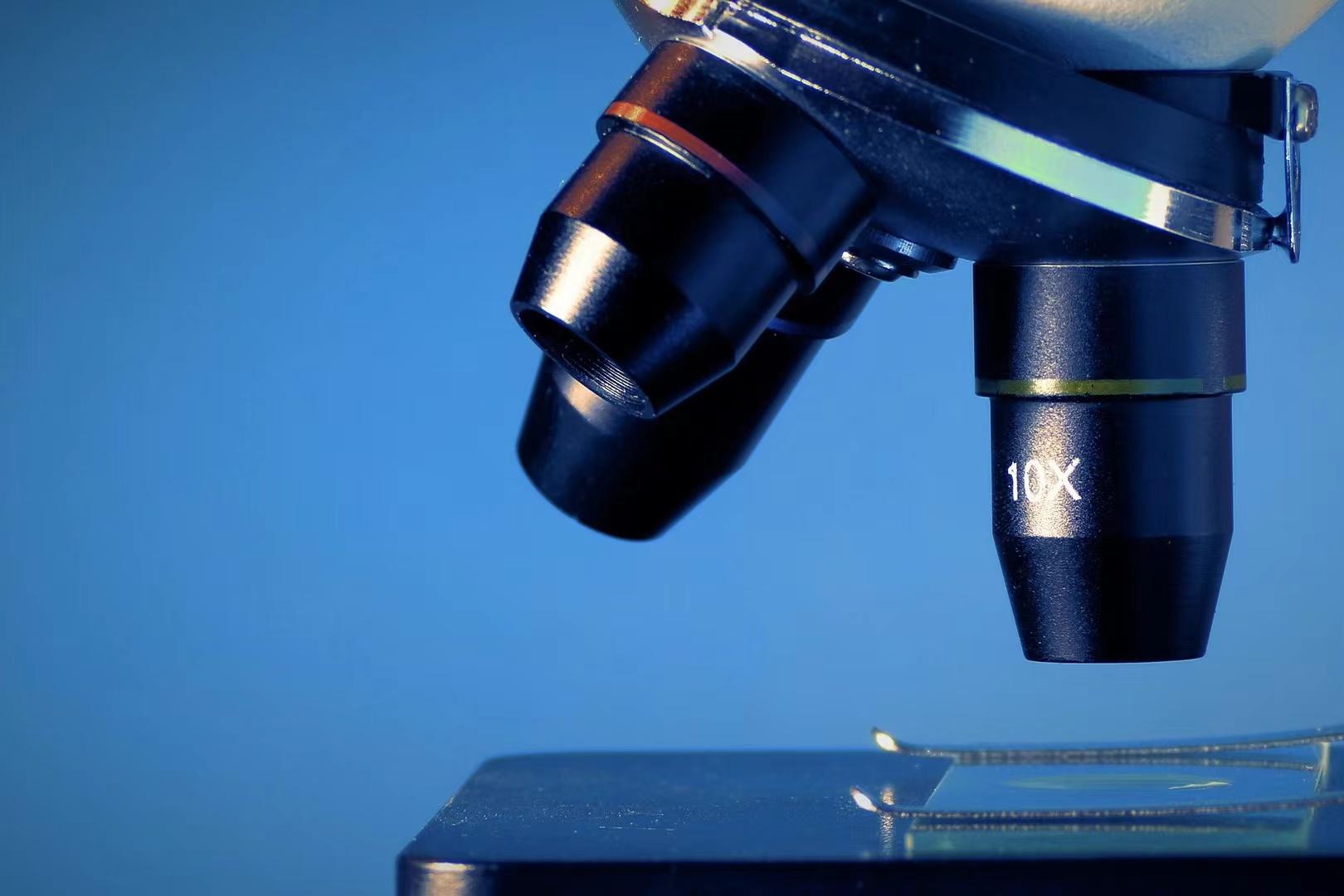
On July 5th, the State Council Information Office held a series of press conferences covering the review and approval of medical devices, medical device regulation, innovative medical devices and other industry issues of concern.

MagBio Genomics said on Monday that it has received US Food and Drug Administration 510(k) clearance, as well as CE marking, for its MagXtract Collection Tube for viral sample collection.
✔ All (3)
✔ Press release (0)
✔ Industry news (3)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
