


Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Themed "Global IVD Innovation & Cross-Regional Integration," this groundbreaking event brings together distinguished experts from around the world to explore the future of diagnostics.
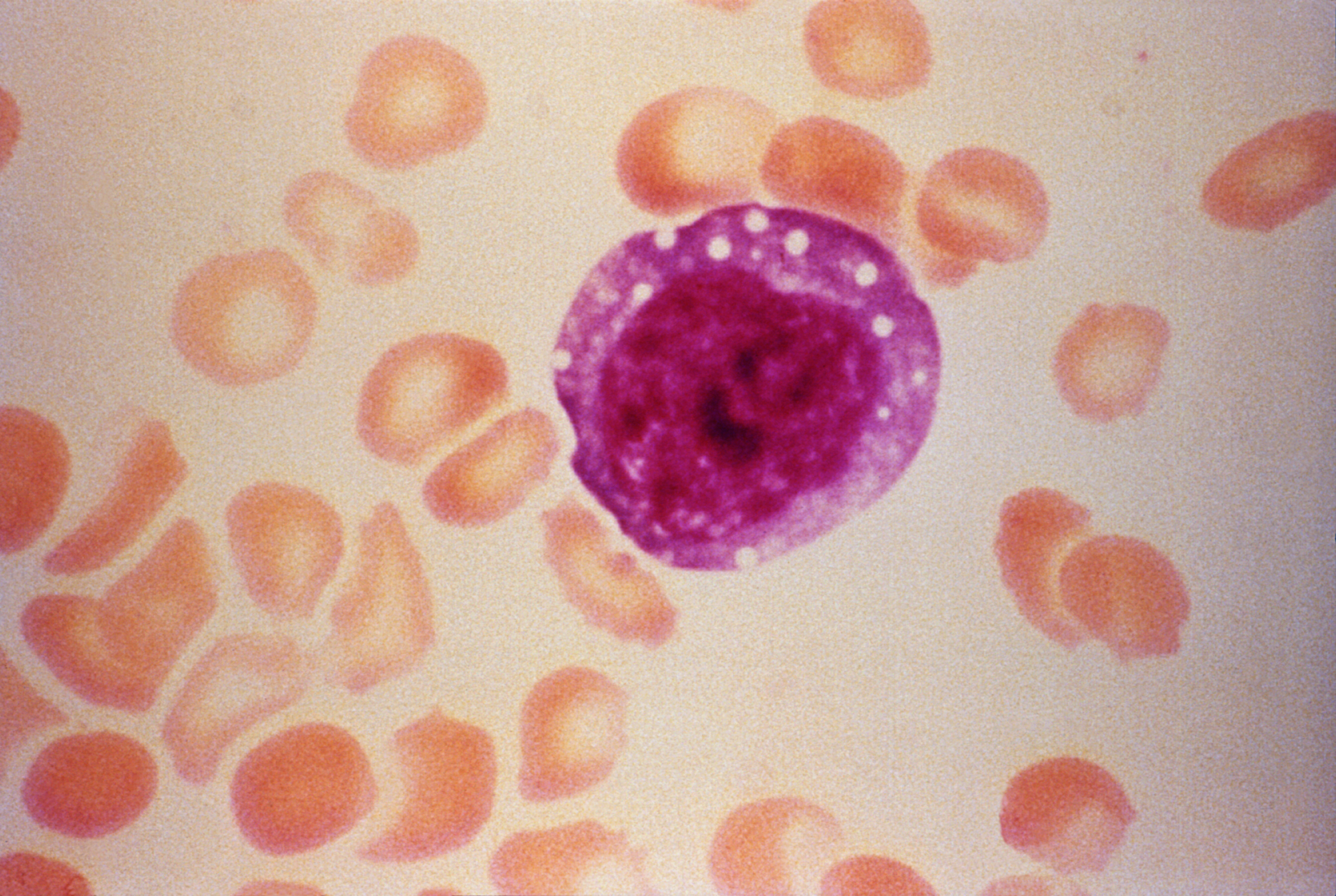
Freenome said on Wednesday that it has partnered with German medical technology firm Siemens Healthineers on the identification of blood-based biomarkers for breast cancer detection.

On 1 December 2021, WHO is calling on global leaders and citizens to rally to confront the inequalities that drive AIDS and to reach people who are currently not receiving essential HIV services.
Abbott and BD contend that they continue to monitor emerging COVID-19 variants of concern, such as omicron, and none have impacted the ability of their respective diagnostics to detect the virus.
French diagnostics firm Novacyt said last week that its Genesig COVID-19 Real-Time PCR assay has been approved in the UK under newly implemented regulations on SARS-CoV-2 tests.

IMPACT trial data shows clear benefit in using Roche's CINtec PLUS Cytology test for women who are at higher risk of developing cervical cancer
Currently available polymerase chain reaction (PCR) tests can detect the emerging Omicron variant of the virus that causes COVID-19, according to the World Health Organization.

The European Union's in vitro diagnostic expert panel has published its first opinion on an In Vitro Diagnostic Medical Devices Regulation (IVDR) submission, delivering a glowing opinion of the filing for a test to screen plasma donor samples for hepatitis E virus (HEV).
The world is in a "race against time" with the Omicron coronavirus variant, said European Commission President Ursula von der Leyen on Sunday, warning during a visit to Latvia that scientists and manufacturers will need weeks to fully understand the new variant.

In order to promote the expansion of high-quality medical resources and balanced regional layout, local governments give full financial support on construction of public hospitals.

One of the first jobs the US Food and Drug Administration’s new inspections council will take on is developing policy around the agency’s use of Remote Regulatory Assessments.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
