


Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Themed "Global IVD Innovation & Cross-Regional Integration," this groundbreaking event brings together distinguished experts from around the world to explore the future of diagnostics.
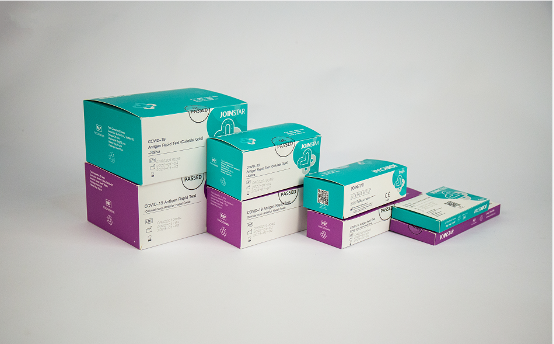
Joinstar’s COVID-19 Antigen Rapid Test (Colloidal Gold) anterior nasal-self testing device, was awarded the CE certificate issued by EU.

Illumina said on Tuesday that it has received CE-IVD marking for its TruSight Oncology Comprehensive (EU) assay this month.

Burning Rock Biotech said on Tuesday that the National Medical Products Administration of China has approved its nine-gene sequencing assay, LungCure CDx, as a Class III medical device.

The development of new scientific ways to see more deeply into the building blocks of nature on a cellular level has led to the some of the greatest advances in medicine over the last century.

Thermo Fisher Scientific Inc., the world leader in serving science, today announced it will invest $97 million to expand its clinical research operations in Richmond, Virginia.
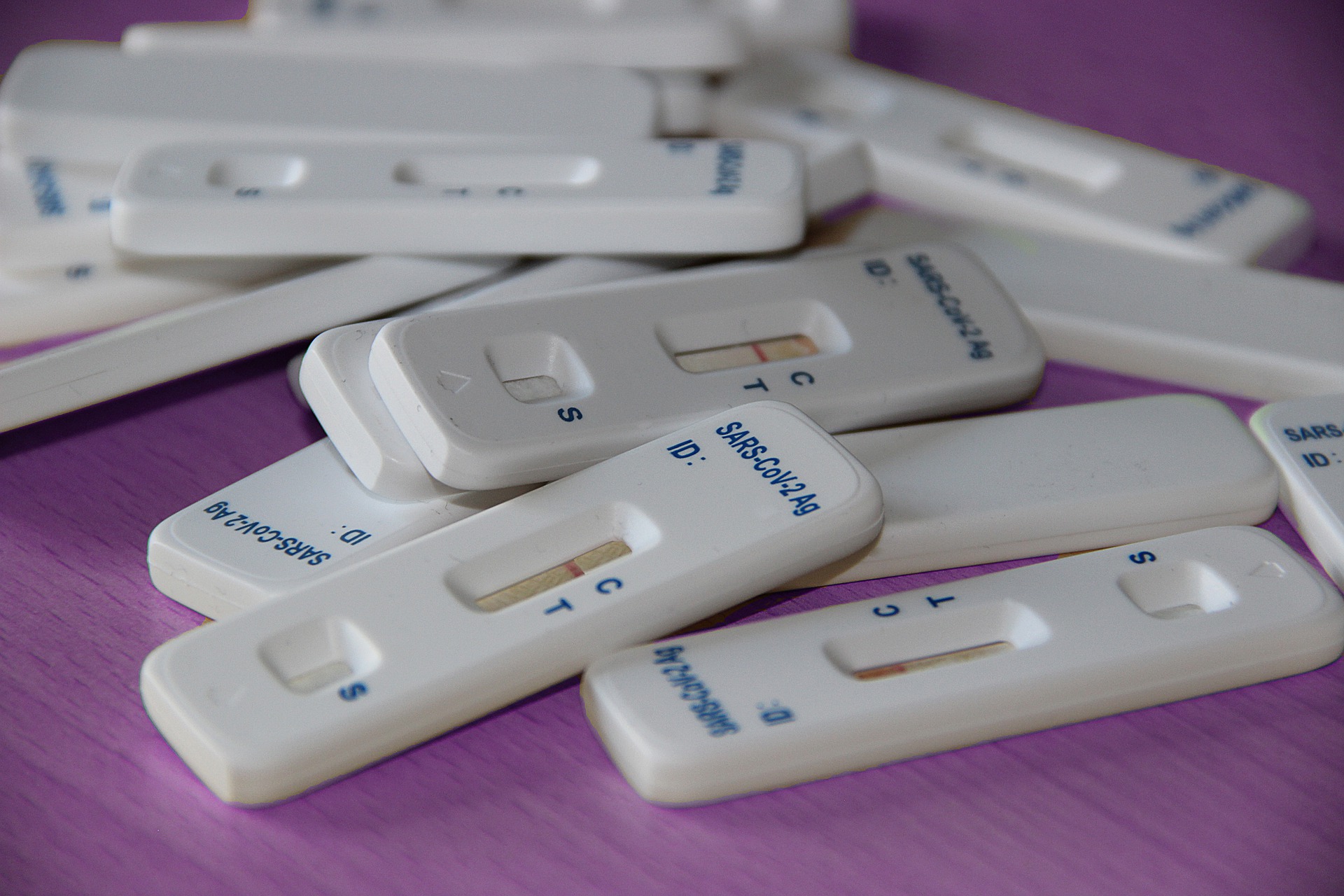
Jilin Province has ordered 12 million antigen detection reagents nationwide in a short period of time, some have been delivered.
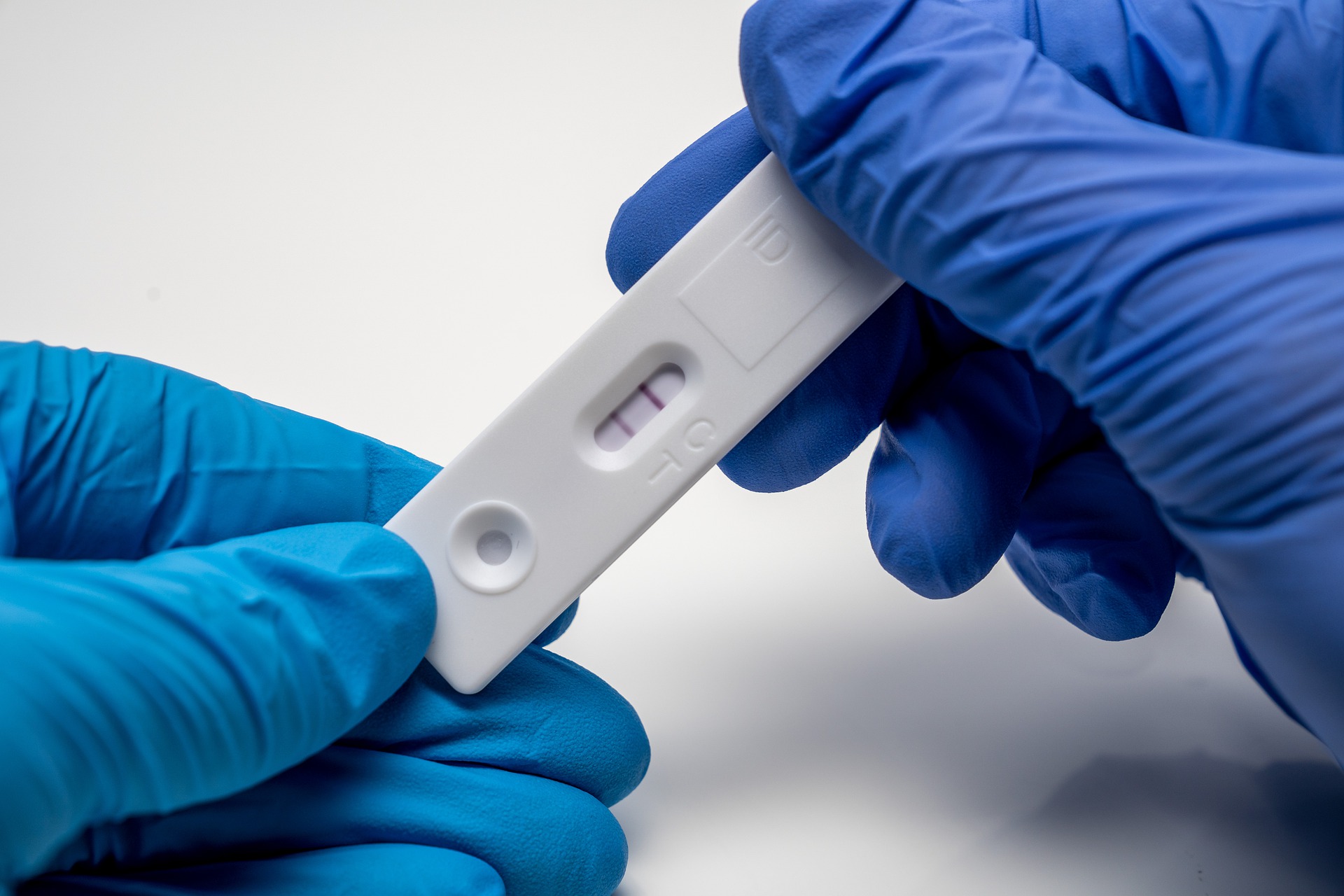
Recently, the domestic epidemic situation is severe, and the release of the "Plan" further optimizes the COVID-19 detection strategy to serve the needs of epidemic prevention and control.
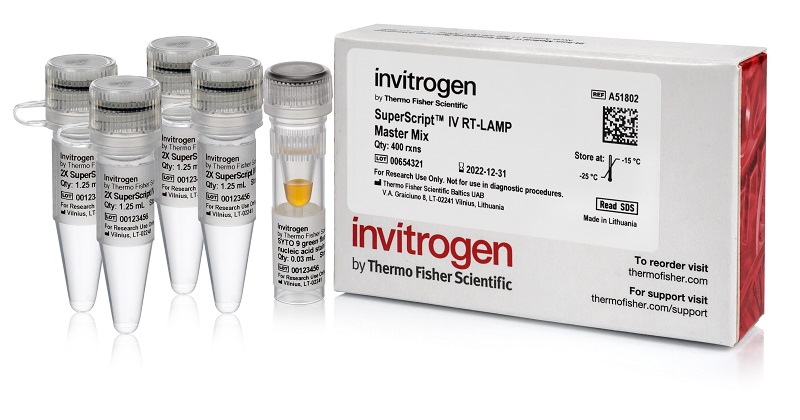
Thermo Fisher Scientific has released the Invitrogen SuperScript IV RT-LAMP Master Mix for the fast and sensitive detection of viral pathogens, including SARS-CoV-2, measles, and influenza.

Lucira Health on Thursday reported a huge year-over-year surge in revenues for the fourth quarter of 2021 on ramped-up manufacture and sale of the company's molecular at-home COVID-19 tests.
Cancer detection is a major public health challenge, and the methods currently available to achieve it, for example MRIs and mammograms, are often expensive and invasive.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
