


Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Themed "Global IVD Innovation & Cross-Regional Integration," this groundbreaking event brings together distinguished experts from around the world to explore the future of diagnostics.

Roche announced on Monday that its Ventana PD-L1 (SP263) assay has received approval from the US Food and Drug Administration for use as a companion diagnostic with Regeneron's Libtayo (cemiplimab) in lung cancer patients.

Recently, U.S. Food and Drug Administration (FDA) and China National Medical Products Administration (NMPA) have released their summaries on medical device innovation in 2022.

The US Food and Drug Administration has allowed InBios International to market its point-of-care anthrax test, the firm announced this week.

BGI Genomics said on Friday that its multiplexed test for SARS-CoV-2, influenza A, and influenza B is now included in the Australian Register of Therapeutic Goods.

Recently, Sansure and Singlera jointly established Jiangsu Yang Shengyuan Biotechnology Co., Ltd. with the registered capital of 10 million CNY at Yangzhou City.

Illumina and Myriad Genetics said on Thursday that they have expanded their strategic partnership on homologous recombination deficiency (HRD) testing.

Biocartis said Thursday that it has secured US Food and Drug Administration 510(k) clearance for a microsatellite instability (MSI) assay to screen for Lynch syndrome in colorectal cancer patients.
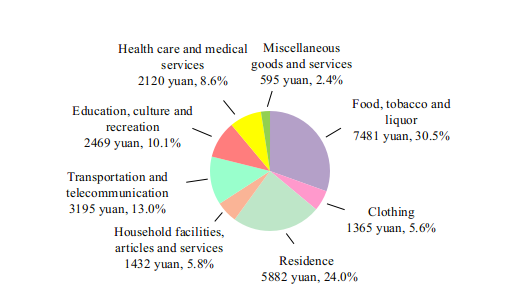
By the end of 2022, there were 1,033,000 medical and health institutions in China, including 37,000 hospitals. The medical and health institutions in China possessed 9.75 million beds, of which hospitals possessed 7.66 million and township health centers had 1.45 million.

QIAGEN N.V. announced the launch of the new QIAseq Platform Partnership program and the first partnership with medical data-analysis provider SOPHiA GENETICSTM that will bring together its QIAseq reagent technology for next-generation sequencing with the SOPHiA DDMTM digital analytics platform.

Agilent Technologies reported after the close of the market that its fiscal first quarter revenues rose 5 percent year over year with gains across all end markets.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
