


To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Invivoscribe is excited to announce that the LeukoStrat CDx FLT3 Mutation Assay has been approved by the U.S. Food and Drug Administration (FDA) to aid in the selection of patients with newly diagnosed FLT3-ITD positive acute myeloid leukemia (AML) who may be eligible to receive treatment with Daiichi Sankyo’s VANFLYTA® (quizartinib).

Abbott today announced financial results for the second quarter ended June 30, 2023.

On July 19, Mindray and Beijing Strong Biotechnologies, Inc. (hereinafter referred to as BSBE) held a strategic cooperation signing ceremony at Mindray` s headquarters in Shenzhen.

Next-generation sequencing data analysis platform company One Codex said on Thursday that it has formed a partnership with Swiss clinical product manufacturer 4bases to bring sample-to-report microbiome analysis solutions to clinicians in the EU.

Siemens Healthineers said Thursday that it has received US Food and Drug Administration clearance for an analyzer designed for immunoassay and clinical chemistry testing in low- to medium-volume laboratories.

Thailand is a country with a high prevalence of thalassemia and sickle-cell anemia, and the related hemoglobinopathies have become a prominent problem in Thailand's public health.

Home health testing company SiPhox Health announced on Wednesday that it has raised $10 million in a seed financing round and $17 million in a Series A round.

AACC has announced that it is changing its name to the Association for Diagnostics & Laboratory Medicine (ADLM).
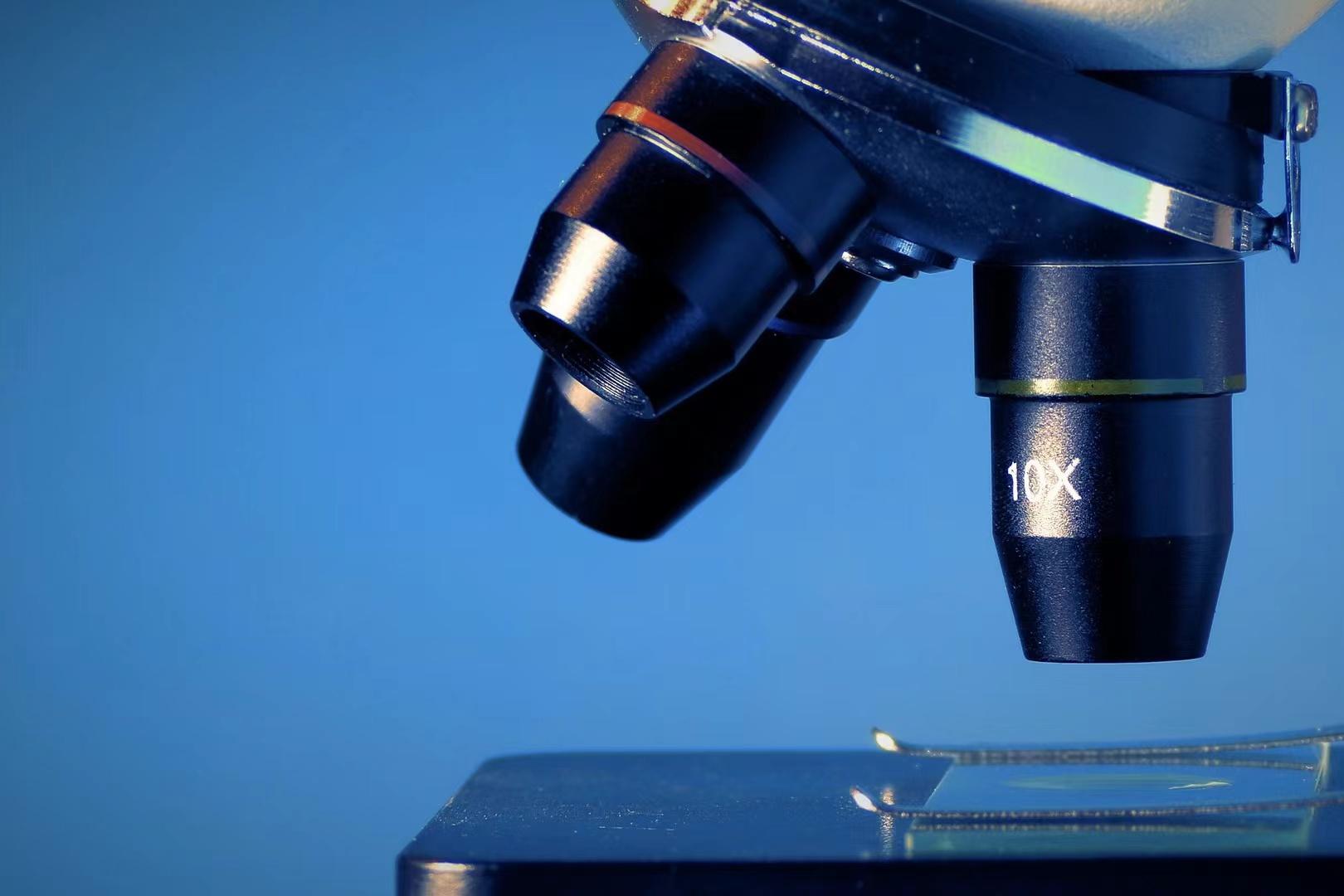
On 5 July 2023, National Medical Products Administration announced the imported Class I medical device product in June 2023. A total of 145 product information, including 7 products in the IVD industry, were finally released.

Co-Diagnostics announced on Tuesday that it has been awarded two grants from the Bill and Melinda Gates Foundation totaling $2.3 million for its tuberculosis and human papillomavirus tests.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
