


To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Thermo Fisher Scientific and Boehringer Ingelheim said on Thursday that they have partnered to develop companion diagnostic tests to help identify patients with non-small cell lung cancer (NSCLC) with specific mutations who may benefit from emerging targeted therapies.

Group sales grow by 1% at constant exchange rates (CER) in the first nine months, showing a strong increase of 7% in the third quarter.

Oxford Nanopore Technologies PLC (LSE:ONT) has secured a £70 million investment from French in-vitro diagnostics (IVD) company bioMérieux, in exchange for a 3.5% stake.
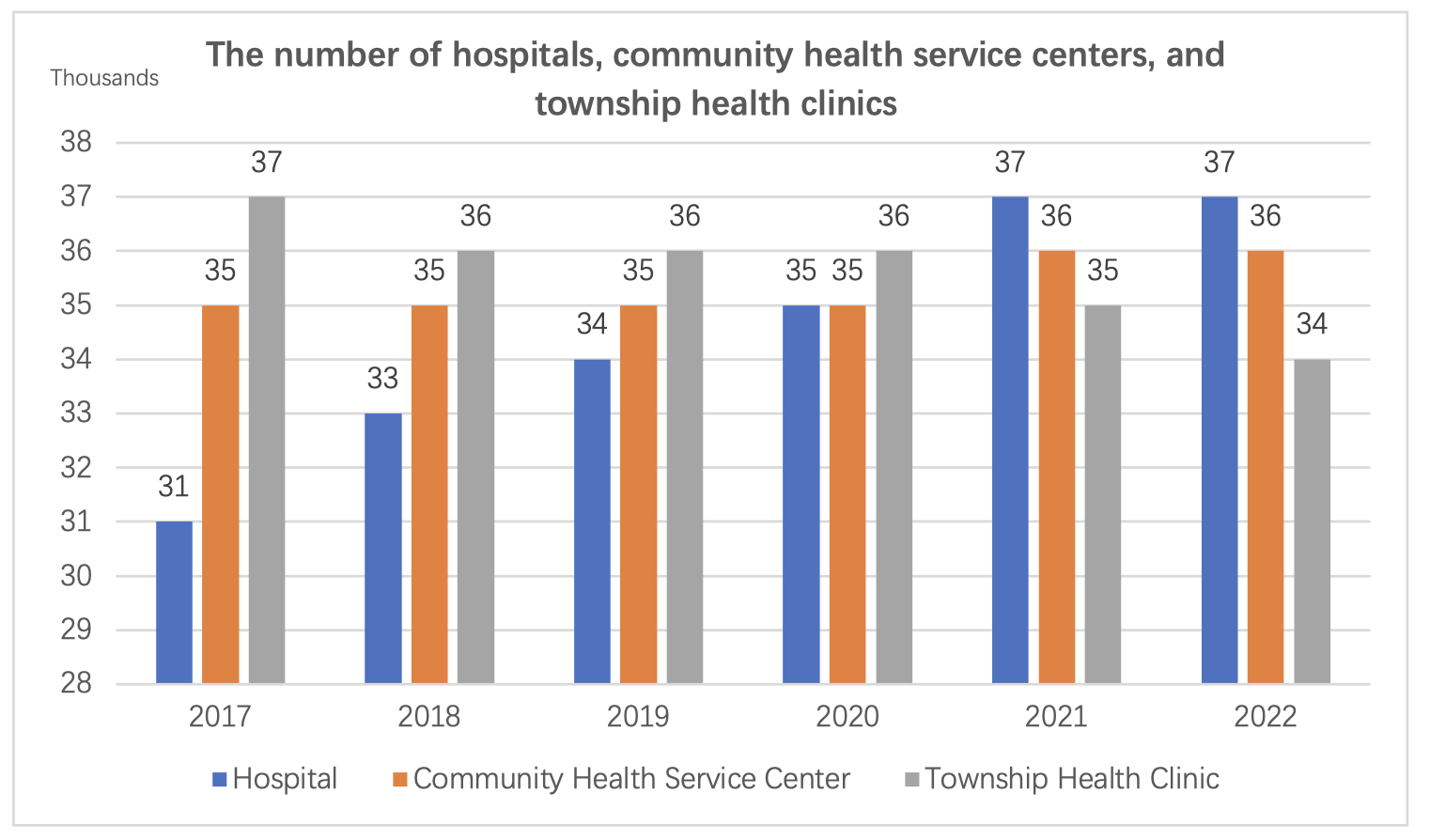
By the end of 2022, the total number of medical and health institutions nationwide reached 1,032,918, an increase of 1,983 compared to the previous year.

Revvity and Danaher subsidiary Sciex said Tuesday that they have signed a distribution agreement that will improve the accuracy of mass spectrometry-based disease screening in newborns by combining Sciex's mass spectrometry instruments and Revvity's reagents.

Abbott (NYSE: ABT) today announced financial results for the third quarter ended Sept. 30, 2023.

As of October 12, 2023, a total of 231 breakthrough medical devices have been approved in China, including 24 IVD products.
Werfen has received 510(k) clearance from the US Food and Drug Administration (FDA) for its Aptiva Connective Tissue Disease (CTD) Essential reagent.

Thermo Fisher Scientific Inc. (NYSE: TMO) (“Thermo Fisher”), the world leader in serving science, and Olink Holding AB (publ) (“Olink”) (Nasdaq: OLK), a leading provider of next-generation proteomics solutions, today announced that their respective boards of directors have approved Thermo Fisher’s proposal to acquire Olink for $26.00 per common share in cash, representing $26.00 per American Depositary Share (ADS) in cash.

China Association of In Vitro Diagnostics (CAIVD) and In Vitro Diagnostics Society of China Association for Medical Devices Industry jointly compiled this report.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
