


To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Today GE HealthCare (Nasdaq: GEHC) announced the release of a new, all-in-one platform of artificial intelligence (AI) apps to support clinicians with breast cancer detection and improved workflow productivity called MyBreastAI Suite.

Quest Diagnostics (NYSE: DGX), the world's leading provider of diagnostic information services, and Scipher Medicine, a precision immunology company, today announced a multi-pronged collaboration designed to expand patient access to diagnostic services advancing precision medicine for rheumatoid arthritis (RA).

China's in vitro diagnosis began in the 1980s. In the past years, the Chinese IVD industry has been following the trend of national reform and opening up and has made great achievements in the world. At present, China's IVD has developed into a complete industrial chain from reagent instrument system to raw materials and from the domestic market to the global market. Its products have covered all the subdivided fields of the industry and become one of the countries with the most complete product lines of IVD industry in the world.

Revvity said on Monday it has received a CE-IVD mark for a molecular testing platform that can be used to screen newborns for spinal muscular atrophy and severe combined immunodeficiency.

Roche announced on Monday that its hepatitis B immunoassay has received CE marking and is available for use in countries accepting the regulatory designation.

Stay up-to-date with the latest happenings in the rapidly evolving field of In Vitro Diagnostics (IVD) in China.

Broncus has entered an equity transfer agreement to acquire a 100% equity interest in Chinese device company Hangzhou Jingliang.

According to the Global Burden of Disease Study 2019, the number of people diagnosed with dementia is expected to almost triple by 2050.

Asep Medical Holdings Inc. ("Asep" or the "Company") (CSE: ASEP) (OTCQB: SEPSF) (FSE: JJ8) is very pleased to announce that, through its subsidiary Sepset Biosciences Inc. ("Sepset"), it has signed a definitive joint venture agreement (the "JV Agreement") with leading Chinese medical diagnostic company, Sansure Biotech Inc. ("Sansure"), through its subsidiary, Hunan Xiang Jiang Sansure Biotech Fund, L.P. (the "Sansure Fund"). Sansure Fund is an investment fund formed by Sansure, Changsha Sanway Spring Venture Capital CO., Ltd. ("Sanway Spring") and certain other investors. The JV Agreement was signed on October 27, 2023.
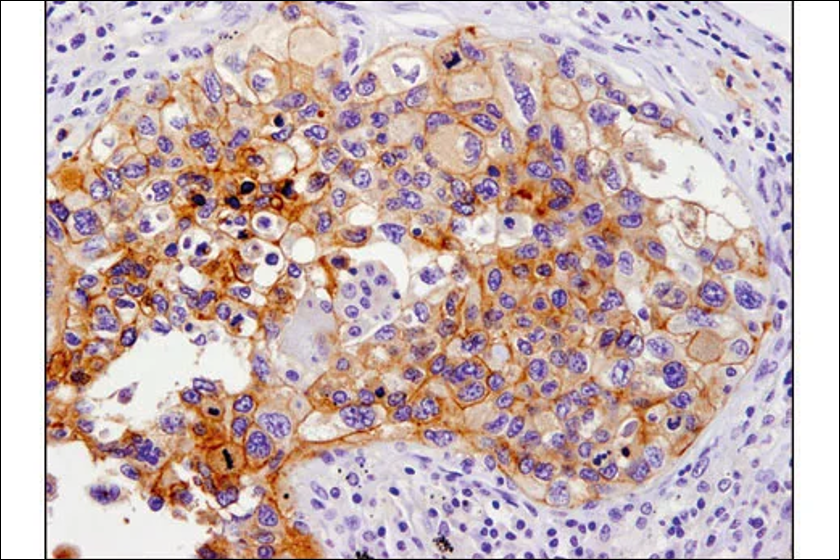
Cell Signaling Technology (CST), a life science biotechnology company and leading provider of antibodies, kits, and services, and Amoy Diagnostics Co., Ltd. (AmoyDx), a China-based innovative molecular diagnostics company in the field of precision oncology, announced today the expansion of their ongoing partnership for companion diagnostic (CDx) development in China.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
