


To ensure the quality and safety of medical services in primary healthcare institutions, China’s National Health Commission (NHC) and other relevant authorities issued a notice on 1 April 2026, outlining a three-year nationwide action.

Microplate-based automatic blood grouping analyzer Metis of Aikang MedTech Co., Ltd. (henceforth referred to as Aikang) received approval for marketing in 2015; and its MGT-based automatic blood grouping analyzer Aigel received approval for marketing in 2016.

Enable Biosciences said on Tuesday that it has received a $3 million Phase IIB Small Business Innovation Research grant from the National Institute of Diabetes and Digestive and Kidney Diseases for its type 1 diabetes test.

The US Food and Drug Administration announced on Tuesday that it has approved AutoGenomics' test to determine whether a patient has an elevated risk of developing opioid use disorder (OUD).

QuidelOrtho Corporation (Nasdaq: QDEL) (“QuidelOrtho”), a global provider of innovative in vitro diagnostic technologies designed for point-of-care settings, clinical labs and transfusion medicine, has received 510(K) clearance from the U.S. Food and Drug Administration for its innovative Savanna PCR platform and Savanna HSV 1+2/VZV in vitro diagnostic test for the detection and differentiation of Herpes simplex virus type 1 (HSV-1), Herpes simplex virus type 2 (HSV-2) and Varicella-zoster virus (VZV) nucleic acids isolated and purified from swabs obtained from cutaneous or mucocutaneous lesion specimens obtained from symptomatic patients.

CACLP's initiative to launch an English/Chinese webinar for non-EU IVD players in next January is set to provide a crucial platform for understanding EU IVDR registration requirements.
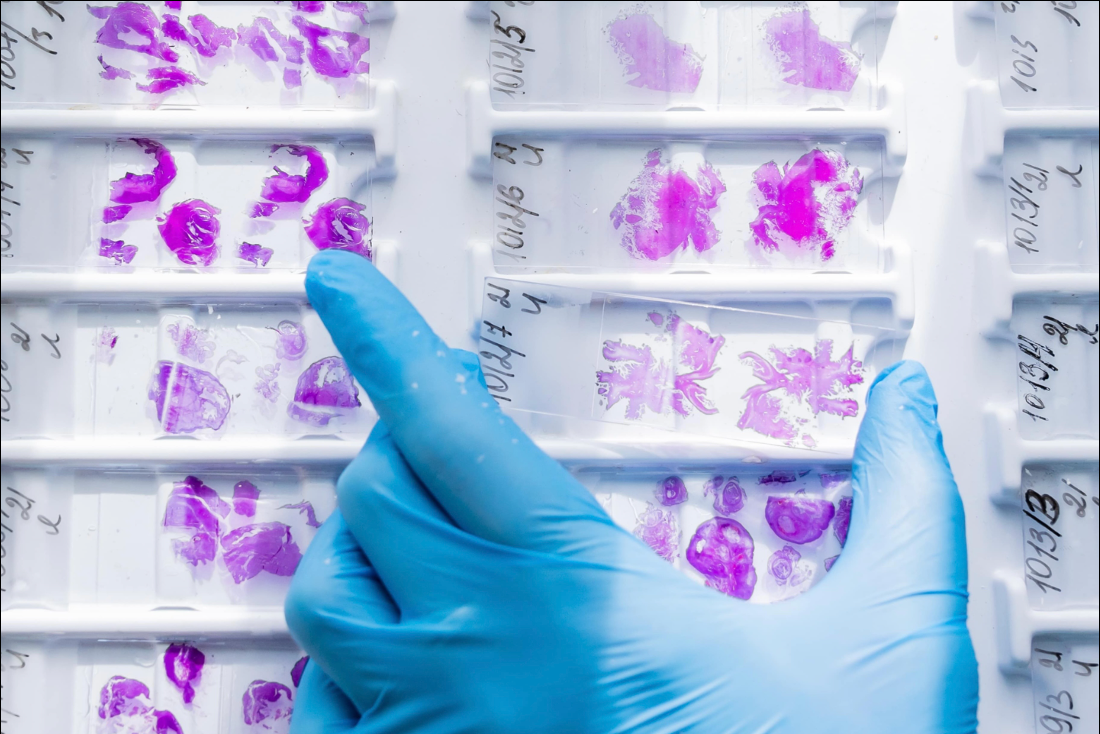
Owkin announced on Tuesday that it entered into a collaboration with MSD — known as Merck in the US and Canada — to develop and commercialize microsatellite instability high (MSI-H) diagnostics for four types of cancer.

Significant regulatory changes across numerous medical fields are coming in 2024 with pharmaceutical and medical device companies urged to be ready for a year of legal alterations.

The immunoassay market has been the main driven force for the development of the in vitro diagnostics industry in recent years.

Euroimmun, a Revvity company, and Xpedite Diagnostics have partnered to offer DNA extraction protocols for point-of-care diagnostics.

Today, Illumina, Inc. (NASDAQ: ILMN) announced that the company will divest GRAIL.

Stay up-to-date with the latest happenings in the rapidly evolving field of In Vitro Diagnostics (IVD) in China.
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
