
Recently, Autobio's hepatitis B virus surface antigen (HBsAg) detection kit (magnetic particle chemiluminescence method) successfully passed the European clinical assessment and obtained the first EU CE IVDR Class D certification in China.
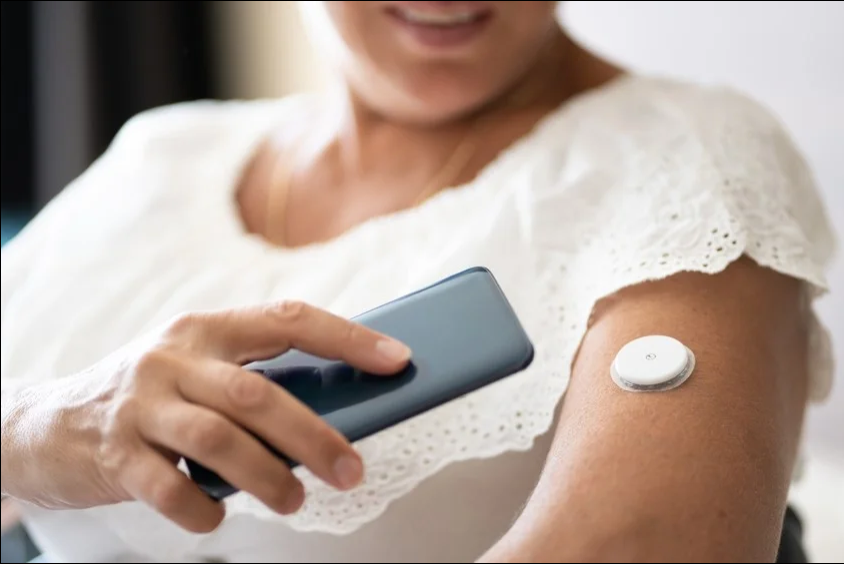
Medtronic has received a CE mark for its continuous glucose monitoring (CGM) device, Simplera, allowing the device to be marketed in the European Union.

Geneseeq Technology Inc. has announced that three of our next-generation sequencing (NGS)-based cancer genetic testing kits, GENESEEQPRIME NGS Tumor Gene Detection Kit (GeneseeqPrime ™), GENESEEQ Homologous Recombination Deficiency Detection Kit (GeneseeqPrime™ HRD), and GENESEEQ Blood Cancer Gene Detection Kit (Hemasalus™ DNA/Hemarna™ RNA), have obtained the European Union’s CE Mark approval.

Recently, Ustar and Hangzhou Biotest Biotech received the IVDR CE certificate from TÜV SÜD, a notified body designated under the EU's IVDR.

DiaSorin said Tuesday that it has secured CE marking for an immunodiagnostic assay to aid diagnosis of severe conditions including sepsis, septic shock, kidney diseases, and lower respiratory and urinary tract infections, and the firm is launching the assay in Europe.

Cepheid announced that it has received the CE mark for Xpert NPM1 Mutation, a molecular in vitro diagnostic test for the quantification of mutant NPM1 mRNA transcripts (types A, B and D in exon 12) in peripheral blood specimens from patients with Acute Myeloid Leukemia (AML).

MGI Tech said last week that it has signed the EU Declaration of Conformity for its DNBSeq-G99 sequencer, granting the instrument CE marking under Europe's new In Vitro Diagnostic Regulation (IVDR).

BGI Genomics said on Friday that its multiplexed test for SARS-CoV-2, influenza A, and influenza B is now included in the Australian Register of Therapeutic Goods.

DiaSorin said on Friday that its second quarter revenues rose 20 percent year over year, driven by strength in molecular diagnostic test sales.
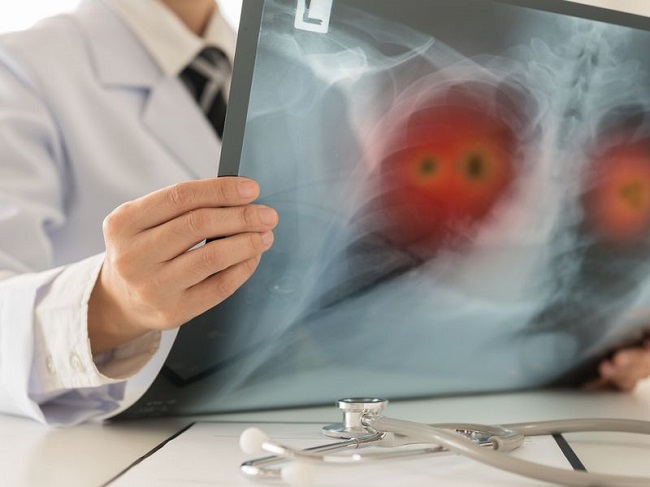
Agilent Technologies on Tuesday announced that it has received CE marking for the expanded use of its PD-L1 IHC 22C3 pharmDx companion diagnostic assay in patients with non-small cell lung cancer.
✔ All (53)
✔ Press release (0)
✔ Industry news (53)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
