
Stay up-to-date with the latest happenings in the rapidly evolving field of In Vitro Diagnostics (IVD) in China.
On 27 October, Ministry of Finance of the People’s Republic of China and National Health Commission of the People’s Republic of China issued a notice.

China Association of In Vitro Diagnostics (CAIVD) and In Vitro Diagnostics Society of China Association for Medical Devices Industry jointly compiled this report.
Listed companies with main IVD business in China have completed the disclosure of their financial results in the first half of 2023.

Foundation Medicine announced on Monday that it is expanding its partnership with Sequanta Technologies to provide its genomic profiling tests for research and oncology therapy development in China.
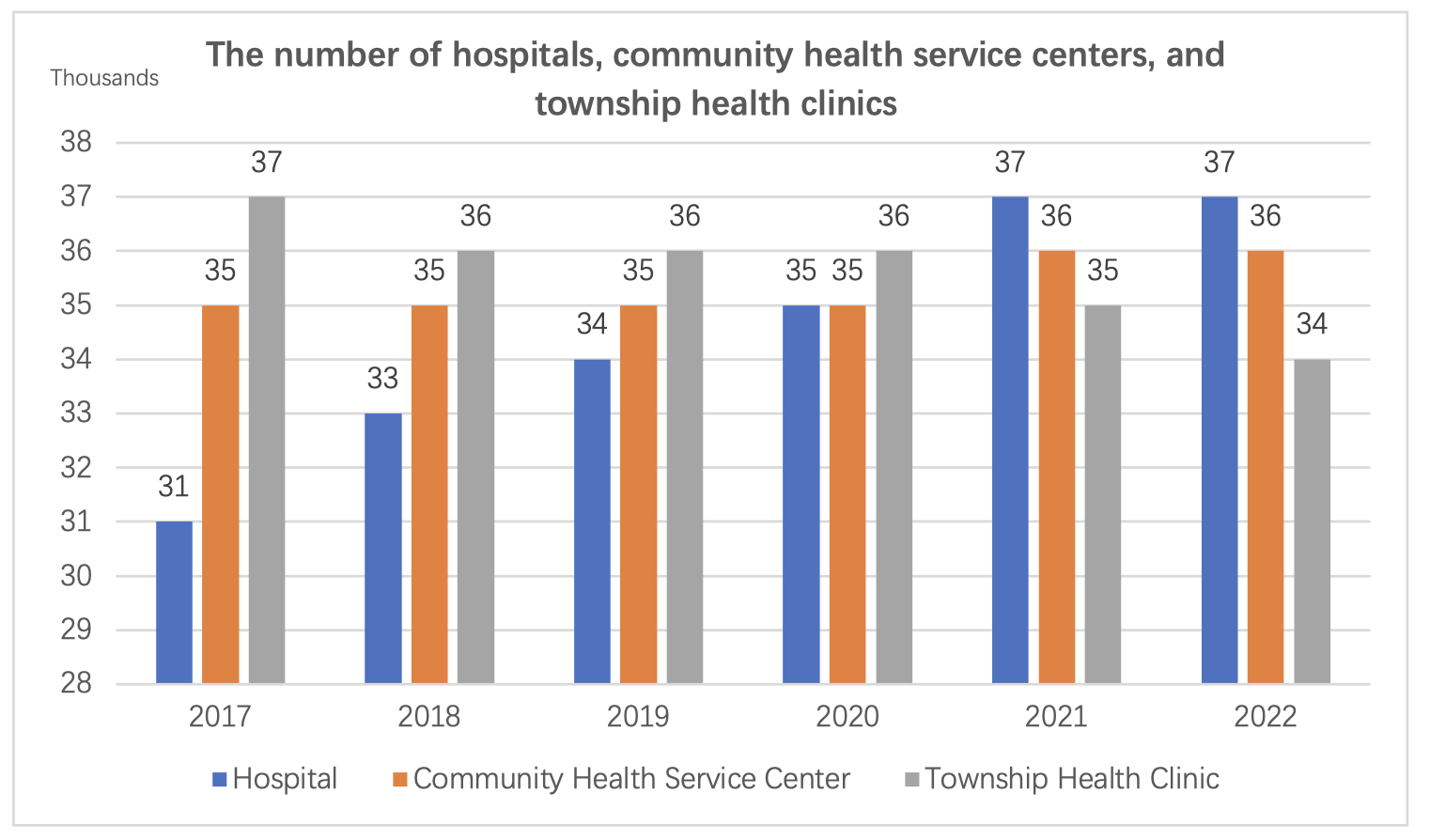
By the end of 2022, the total number of medical and health institutions nationwide reached 1,032,918, an increase of 1,983 compared to the previous year.

China Association of In Vitro Diagnostics (CAIVD) and In Vitro Diagnostics Society of China Association for Medical Devices Industry jointly compiled this report.

Stay up-to-date with the latest happenings in the rapidly evolving field of In Vitro Diagnostics (IVD) in China.

Recognizing the long-term needs of the biopharma industry in China and Asia-Pacific, global life sciences leader Cytiva has opened its expanded Shanghai facility. The facility is 11 000 m2, fully operational, and designed to help advance and accelerate the development of novel therapeutics. Customers have access to a wide range of technologies, solutions, and services, including Cytiva’s Fast Trak training center, a Center of Excellence for single-use technologies, and a new automation and digital lab. It is also home to China’s online experience center, customized bioprocess solutions labs and compliance validation labs.

For those international medical device companies aiming to localize in China, what exactly does "manufactured within China" mean? To what extent does "localization" qualify as "manufactured within China"?
✔ All (69)
✔ Press release (3)
✔ Industry news (66)
Don’t miss important updates about the show and the in vitro diagnostic industry.
Sign-up for our newsletter today.

Copyright © 2026 GL events Ruihe (Shanghai) Exhibition Co., Ltd. All Rights Reserved. ( 沪ICP备12004745号-1 )

We deliver the latest IVD news straight to your inbox. Stay in touch with CACLP News.
sign-up for our newsletter today.



To ensure our newsletter hit your inbox, make sure to add @caclp.com to your safe senders list. And, as always, feel free to contact
us with any questions and thanks again for subscribing.
 Go back
Go back
